Long-term implants and single-use DEVICEs
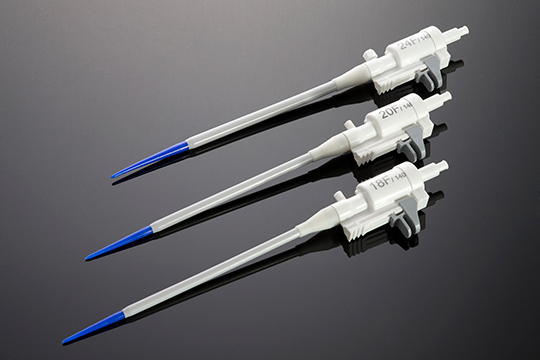
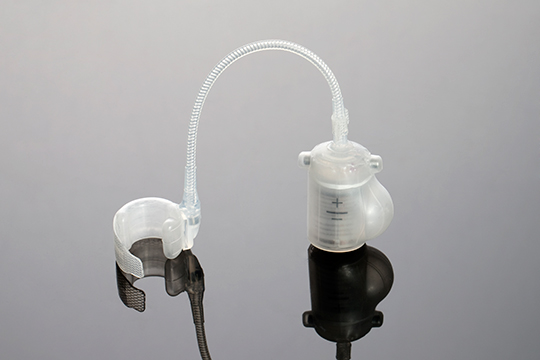
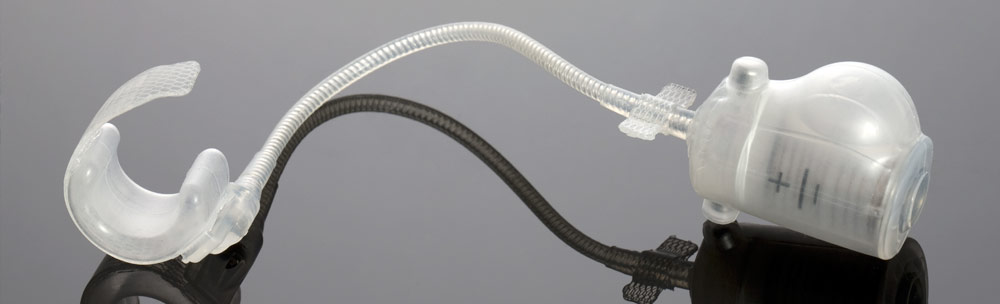
Over the years, STATICE has shown and demonstrated their ability to design and develop a selection of long-term implants and single-use devices in classes I to III, in a number of industries ranging from neurology to cardiology and ophthalmology, ENT, obesity surgery, and others.
STATICE will remain close to you throughout the design and manufacture of your medical device, all the while considering the technical aspects or constraints together with the required specifications:
- Selection and choice of biocompatible materials (silicon, bioresorbable thermo-plastics) ISO 10993
- Procedures for the transformation of materials (LSR injection, plastic injection, extrusion, machining etc...)
- Validation of any special requirements or conditioning (gluing, welding, clipsing etc...) and validation of required product performance
- Ergonomics, facility of use
- Application of all applicable Standards required and reference 93/42/CE.